|
2/24/2024 0 Comments Periodic table molar mass rounded 
The former definition of the mole was that a mole was 12 g of 12C contains 1 mole of 12C atoms (its molar mass is 12 g/mol). 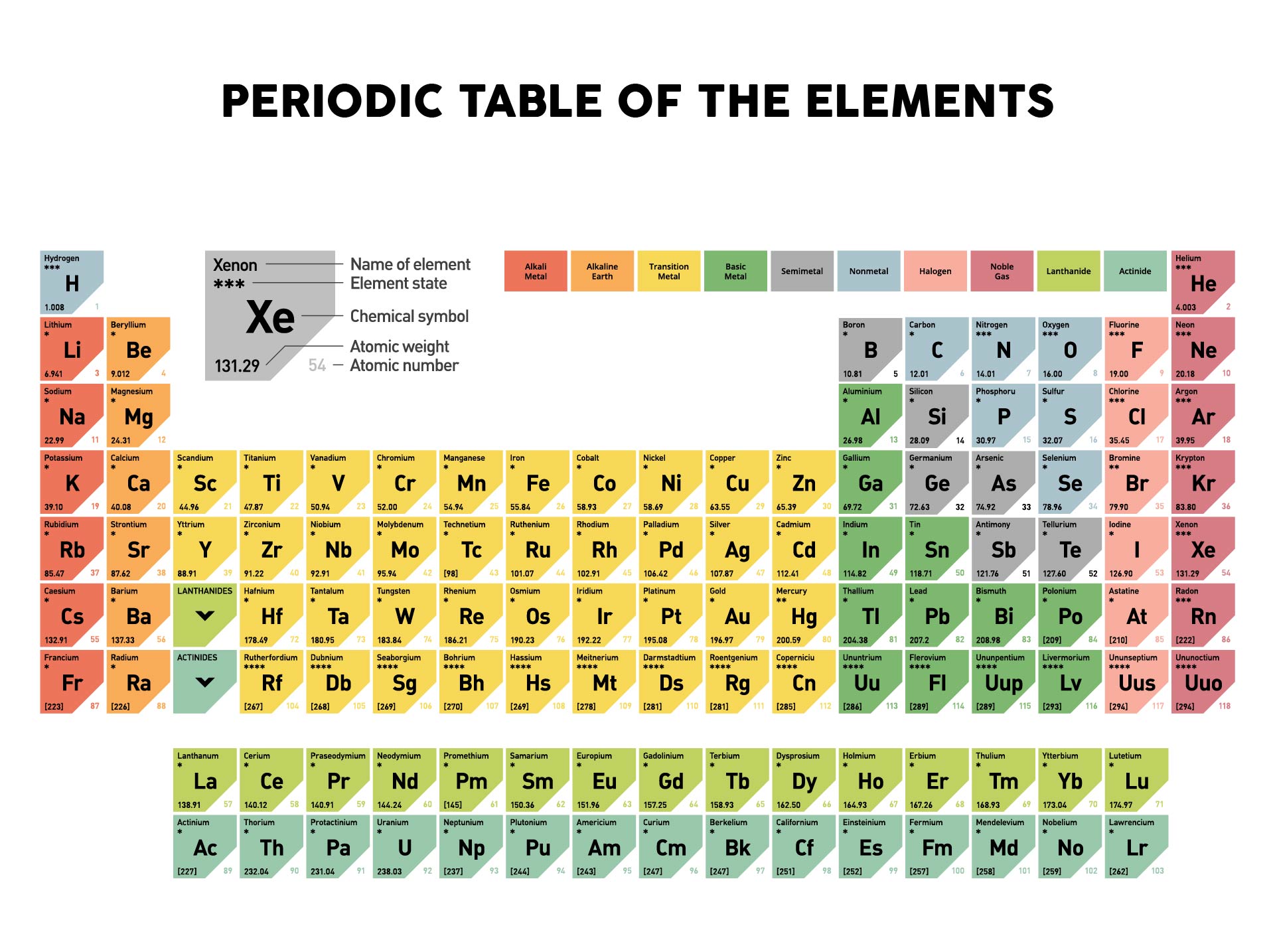
Per the amu definition, a single 12C atom weighs 12 amu (its atomic mass is 12 amu). Periodic Table: Representative Elements & Transition Metals 7m. The molar mass is an average of many instances of the compound, which often vary in mass. 1 The molar mass is a bulk, not molecular, property of a substance. (credit: modification of work by Mark Ott).īecause the definitions of both the mole and the atomic mass unit are based on the same reference substance, 12C, the molar mass of any substance is numerically equivalent to its atomic or formula weight in amu. In chemistry, the molar mass ( M) of a chemical compound is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of said compound. Since many elements have a number of isotopes, and since chemists rarely work with one atom at a time, chemists use average atomic mass. 
From left to right (bottom row): 32.1 g sulfur, 28.1 g silicon, 207 g lead, and 118.7 g tin. What is the average atomic mass of neon Round your answer to three decimal places., Atoms of the same element can have different: atomic numbers numbers of protons mass numbers none, Which element weighs, on average, approximately one. From left to right (top row): 65.4 g zinc, 12.0 g carbon, 24.3 g magnesium, and 63.5 g copper. Study with Quizlet and memorize flashcards containing terms like Out of 200 neon atoms, 170 will be neon-20(19.992 amu) and 30 will be neon-22(21.991 amu). You will need to refer to a periodic table for proton values.\) atoms -1.00 mol of atoms. In this notation, the atomic number is not included. A row on the periodic table is called a period. The atomic mass of tin (Sn) is 118. Heres a nice Periodic Table and more pdf for you to use for this class. The atomic mass of each element is found under the element symbol in the periodic table. 
Every hydrogen atom has one proton in its nucleus. The periodic table is your ultimate conversion chart for converting any substance into another substance and doing so with exact proper amounts (masses and moles). Converting the mass, in grams, of a substance to moles requires a conversion factor of (one mole of substance/molar mass of. If the mass of a substance is known, the number of moles in the substance can be calculated. Every hydrogen atom has one proton in its nucleus. Hydrogen, at the upper left of the table, has an atomic number of 1. In this table, an elements atomic number is indicated above the elemental symbol. The Periodic Table with Atomic Mass will give you the atomic weight. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. Hydrogen, at the upper left of the table, has an atomic number of 1. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol). The mass number is established by rounding the atomic weight to the nearest whole number. In this table, an element's atomic number is indicated above the elemental symbol. Basically, I first wrote down the amount in grams, and I used the molar mass of Carbon (which can be found on the periodic table under Carbon) 12.01 g/mol to convert 4.01 g of Carbon to moles of Carbon. How can I find the molar mass of an element The molar mass of elements is found by looking at the atomic mass of the element on the periodic table. It is also sometimes called: Molecular Mass, Molecular Weight, Formula Mass, or Formula Weight. The atomic mass of a solitary atom is its absolute mass and is regularly expressed in atomic mass units or amu. Symbol-mass format for the above atom would be written as Cr-52. The periodic table (see figure below) displays all of the known elements and is arranged in order of increasing atomic number. One atomic mass unit (u) is equal to 1/12 the mass of one atom of carbon-12. For an example of this notation, look to the chromium atom shown below:Īnother way to refer to a specific atom is to write the mass number of the atom after the name, separated by a hyphen. The mole concept can be extended to masses of formula units and molecules as well. According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. The "A" value is written as a superscript while the "Z" value is written as a subscript. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Both the atomic number and mass are written to the left of the chemical symbol. The composition of any atom can be illustrated with a shorthand notation called A/Z format. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
